Everything about Lars Fruergaard Jørgensen is understated. The chief executive of Danish drugs group Novo Nordisk grew up on a pig farm in Jutland and from an early age was expected to muck out the animals.
While most of his rivals take private jets to their appointments, he waits for commercial connections. Tall, thin and thoughtful, Jørgensen is far from the conventional image of a swashbuckling, dealmaking CEO: he started his career as an economist in the healthcare and planning department.
But in his low-key way, Jørgensen is pioneering a commercial innovation that could have a profound impact not just on healthcare, but on societies, on public finances and on our relationship with food.
Novo Nordisk is the company behind the first game-changing treatments that are used for obesity: Wegovy and Ozempic. Before these drugs, the only truly effective treatment was bariatric surgery, which is expensive and sometimes risky. Now there is a simple injection.
A safe and readily available treatment for obesity could have a huge impact on human health, while also generating savings on treating other diseases. Obesity will affect an estimated 1bn people by 2030 — with all the associated impact that has on rates of diabetes, heart disease and mobility. In the US alone, the economy loses up to $30bn a year in sick days due to the condition, according to research from Cornell.
And it is not just obesity: there is now good evidence to suggest the company’s drugs could also help prevent heart attacks and there is even some hope they could be used to treat Alzheimer’s by reducing inflammation in the brain.
The Novo Nordisk drugs have been several decades in the works and Wegovy was first approved in the US in 2021. But this is the year that the momentum behind them has become unstoppable, when it became clear that they are more than just a celebrity fad.
Barclays forecasts sales of Wegovy, which is used for obesity, to hit $4.2bn this year and $7.3bn next, with Ozempic, designed for diabetes but often prescribed off-label for obesity, predicted to hit $13.3bn in 2023 and $16.5bn in 2024.
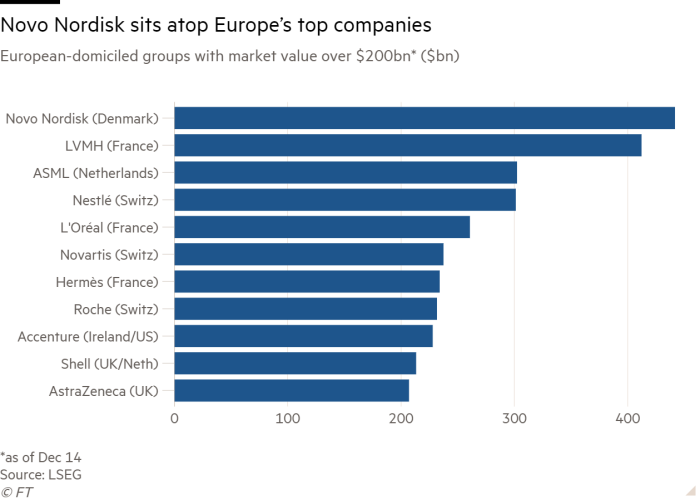
The success has turbocharged Novo Nordisk: with little fanfare, it has become the most valuable company in Europe, overtaking luxury group LVMH, maker of Louis Vuitton and Moët & Chandon.
For his role conducting this model of patient, persistent but transformational innovation, the Financial Times has chosen Jørgensen as its Person of the Year.
Jørgensen, who drives himself to work in an electric car and goes kayaking on the lake near his house to reflect, would never be as brash as to claim his product can be a panacea for society’s ills — in the style of some Silicon Valley executives. But in his typically earnest way, he insists that the company and the industry can have a “huge chance of impacting the world”.
“I feel a great sense of responsibility for actually succeeding together with society,” he tells the FT, “because I think as an industry and as a company, we have part of the key to solving some of the huge societal problems, ageing populations, chronic diseases.”
Novo Nordisk also faces a number of potential obstacles that could still hamper its expected growth. The surge of popularity in the drugs — fuelled in part by slimming celebrities — has caused a supply shortage and created a booming black market. Concerns rumble about side effects. Rival Eli Lilly has won approval for its own obesity drug, which could be more effective and currently has a lower list price.
Most of all, Jørgensen still needs to convince many governments and cash-strapped health systems that it makes sense to pay for the drug now in order to save money later. And that in part requires a shift in mindset so that obesity is considered a disease that can be treated with medicine — like, say, high blood pressure — and not as a personal failing.
The company insists that for all the growing success, it is not getting ahead of itself. According to Kasim Kutay, chief executive of Novo Holdings, which controls the company, and who is a Novo Nordisk board member, Jørgensen reacted with a sense of “immense responsibility” when the company became the largest in Europe.
“It’s not something that is talked about or boasted about as . . . a feat in itself,” he says. “It’s about how do we use our scale and size to deliver on our responsibility to patients?”
Taking the long view
The drugs began life at Novo Nordisk 32 years ago — by coincidence, when Jørgensen joined the company. Wegovy and Ozempic are both made from semaglutide, a version of an appetite-reducing hormone called GLP-1. But in the body, the hormone only lasts for minutes, so Novo’s scientists spent years making it stable enough to use as a medicine.
Jørgensen’s time at the company has coincided with a long-term bet on the potential of this new science. The first real breakthrough came 14 years ago, when the drugmaker got its first approval for a GLP-1 drug for diabetes in 2009. Another version followed in 2015, targeting weight loss. But it only helped patients lose about 5 per cent of their body weight. It would take six more years until Wegovy was approved, after a trial showed an average of 15 per cent weight loss.
Bent Dalager, an old friend of Jørgensen, describes how his childhood on the farm in Jutland made him a patient leader. “The thing about growing is throwing out these seeds, and they will become something later,” he says. “That’s actually a fairly good frame of mind to have if you work in the pharma industry.”
Novo Nordisk could invest for the very long term partly because of its unusual ownership structure. Initially called Nordisk Insulinlaboratorium, the company was founded in 1923 by the Danish Nobel laureate August Krogh, pharmacist August Kongsted and scientist Hans Christian Hagedorn. The Canadian scientists who discovered insulin granted the pair permission to produce it in Scandinavia, with a caveat: the proceeds from its sale should be reinvested in research.
So they set up the Novo Nordisk Foundation, which thanks to the company’s growth is now the world’s largest philanthropic foundation by assets. Novo Holdings, which manages the foundation’s wealth, has 77 per cent of the voting rights of Novo Nordisk.
Martin Jes Iversen, an associate professor of strategy and innovation at Copenhagen Business School, says the structure kept the company committed to its broader purpose beyond profitability.
It also ensured Novo Nordisk was not for sale. “So not even Elon Musk in a wild night could take over or attempt to take over Novo Nordisk,” he says.
Starting at the company in 1991, Jørgensen has worked in the US, Japan and the Netherlands, where he was one of few foreign executives to learn Dutch during his posting.
Marcus Schindler, Novo Nordisk’s chief scientific officer, says Jørgensen is committed to Novo’s “ethical guardrails” and is “uncompromising” about serving patients, for example, insisting the company will never stop producing insulin, even though it is less profitable than other drugs.
Shortly after Jørgensen joined the company’s development committee but before he became chief executive, Novo needed to decide whether to invest more in trials for an earlier obesity drug. Jørgensen says it was a “big decision” because of the potential risks to Novo’s reputation. Most other pharmaceutical companies avoided weight-loss drugs because previous attempts had been dogged by dangerous side effects.
“When you look at the field of obesity medicine, most of them were considered as either not very efficacious, or potentially also with an unattractive safety profile,” he recalls.
This year, Jørgensen says his most important decisions have been about saying no. While the organisation is full of ideas of how to invest its windfall, Jørgensen is committed to doubling down on developing products for obesity, diabetes and cardiometabolic diseases.
“If there’s an unmet need, if it’s something that’s a core capability, I feel very, very comfortable in taking huge risks,” he explains.
Cultural influence
Unsurprisingly, Jørgensen is not on TikTok, where #Ozempic has clocked up 1.3bn mentions. He confesses the company was “late” to understanding the cultural impact of its drugs: it looked for feedback from the clinicians who the industry calls “key opinion leaders” rather than Hollywood influencers.
When Kim Kardashian wore Marilyn Monroe’s dress to the Met Gala last year, she set off rumours about a diabetes drug helping celebrities slim down dramatically. Lauren Sherman, a fashion correspondent at digital media company Puck, remembers it looked like she had lost a lot of weight in the previous two weeks.
By the Met Gala this year, Sherman says the drug had become so prevalent it was an “accessory”, even for some of the thinnest famous people. “I do think that it is causing people to think, ‘If it is accessible and I don’t do it, am I going to look like an idiot?’” she says.
Yet most celebrities, including Kardashian, deny using drugs. Sherman says this is partly because being thin is still associated with signalling that you are a “disciplined” person, who does not “overeat”.
Their use of Ozempic, which has been in greater supply than Wegovy, highlights an inequality: the wealthy can get doctors to prescribe the drug, even if they don’t have diabetes, while some real patients have struggled to get hold of it.
Celebrities’ unofficial promotion has helped create a rush for the drugs, with regulators warning about fake injection pens, and reports that online pharmacies will prescribe it to people so thin they could be suffering from eating disorders. The surge in demand has exacerbated supply shortages partly caused by manufacturing problems.
Now, the executive management team gets updates on how the drugs are trending on social media every week. Jørgensen wants to shift from a social media debate to a medical discussion with doctors about the benefits. “It creates a sense of urgency in the company to make sure that we communicate in the right way,” he says.
A broad impact
The drugs have jolted financial markets as well as parties for the rich and famous. Emily Field, an analyst at Barclays, jumped out of her chair when she saw the initial data from Novo Nordisk’s cardiac outcomes trial in August. Wegovy had shown it could cut the risk of serious cardiac events such as heart attacks by 20 per cent in overweight or obese patients with cardiovascular disease.
It was the first solid evidence that the drug was not just a slimming jab, but a powerful preventive tool. Shares jumped too: up as much as 16 per cent as even generalist investors started to appreciate this was a drug that could transform healthcare.
“It just woke up so many people to the whole story,” says Field. “It was such a game-changer.” Field says the drugs are now rightly seen as a technology with widespread impact, like artificial intelligence. Suddenly, consumer analysts were worrying about how the drug could dampen appetite for products from companies such as doughnut maker Krispy Kreme, while medical technology investors worried it could be the end of bariatric surgery.
The clinical trial was one of the first big decisions Jørgensen made after taking over as CEO in 2017. The move carried substantial risk: it can cost hundreds of millions of dollars to test a drug on 17,000 people and there was no guarantee that the results would demonstrate broad health benefits. Now the bet has paid off, Jørgensen hopes to use the data to convince health systems that the drug will save lives and cut costs.
Fatima Cody Stanford, an expert in obesity medicine at Harvard Medical School, says the drugs will be “highly influential” and may lead to a decline in the need for treatment for conditions such as hypertension, kidney disease, fatty liver disease, diabetes and sleep apnoea.
Novo Nordisk is running a late-stage trial to see if semaglutide could treat the widespread neurodegenerative disease Alzheimer’s, and external researchers are also intrigued about the potential for the drugs to be used to treat alcohol addiction.
But there are still problems to solve. Many patients struggle with side effects of nausea, diarrhoea and constipation, especially in the early stages, clinicians are worried about muscle loss, and the European regulator is investigating if there’s a link between taking the drugs and suicidal thoughts.
For healthcare systems, the biggest concern is that when people stop taking the drugs, they regain weight. Some payers are reluctant to commit to covering a drug for the rest of people’s lives, especially when the potential pool of patients is so vast. Some 42 per cent of the population in the US is obese, and 17 per cent in the EU, while many overweight people could also benefit.
The drugs risk exacerbating health inequities. Wegovy has a US list price of $1,300 a month, though the net cost is usually about half that, and the price in Europe is lower. Without greater insurance coverage in the US, or more widespread availability in European public health systems, they will be out of reach for many of the poorest.
This cost has also raised some questions about the likely cost savings. Airfinity, a health analytics firm, estimated that even after a 65 per cent rebate on the list price, it could cost $1.1mn spent on Wegovy to prevent one heart attack.
Mark Sculpher, director of the centre for health economics at the University of York, says the jury is still out on the long-term effects of the drugs, which could affect their true value. Like most drugs, they were approved on a few years of data. “Pharmaceutical companies are forever promising hugely transformational, innovative products,” he says. “And sometimes they are. But very often they are not.”
Jørgensen believes by getting ahead of disease, governments and insurers can save on the more than 80 per cent of healthcare funding that ends up being spent on chronic conditions.

He is not alone in hoping for a substantial shift towards prevention in healthcare, but systems have struggled to make the change when their tight budgets are stretched across an already sick and ageing population. To try to reach more patients, Novo Nordisk is in talks with health systems about flexible pricing to allow them to defer some of the cost of the drugs until they are seeing benefits.
Jørgensen sees continued innovation as another way to help make the argument. Next year, the company will complete a late-stage trial of its next generation drug CagriSema, which analysts expect to show even more significant weight loss, and report data on an early study of a promising obesity pill. In the long run, he dreams of “vaccine-type interventions” for chronic diseases and is investing in discovering how to prevent obesity in the first place.
Schindler, the chief scientific officer, believes Jørgensen is probably “to some degree, embarrassed” to be chosen as the Person of the Year. “I think Lars would feel that it [the recognition] stands for many other people, it stands for an organisation, it stands for a team,” he says.
Jørgensen says the company has long believed in the importance of the obesity drugs but that the world is only just waking up to their potential impact.
“I have many times over the years said that I believe this has the potential to be one of the most meaningful medical interventions in terms of what it does to individual patients,” Jørgensen says, “but also the contribution it [can make] to healthcare systems.”
Data visualisation by Keith Fray


